Researchers in Japan have come up with a new way to create innovative materials by blending metals with precision control. Their approach is based on a concept known as atom hybridization, and it will open up the doors to an area of chemistry that has been previously unexplored, and it could also lead to the development of advanced functional materials.
Multimetallic clusters, which are usually made of three or more metals- are getting more and more attention as they display the properties that are not something that can be achieved by single-metal materials. If a range of metal elements are freely blended, researchers expect that as-yet-unknown substances can be discovered and highly-functional materials might also be developed.
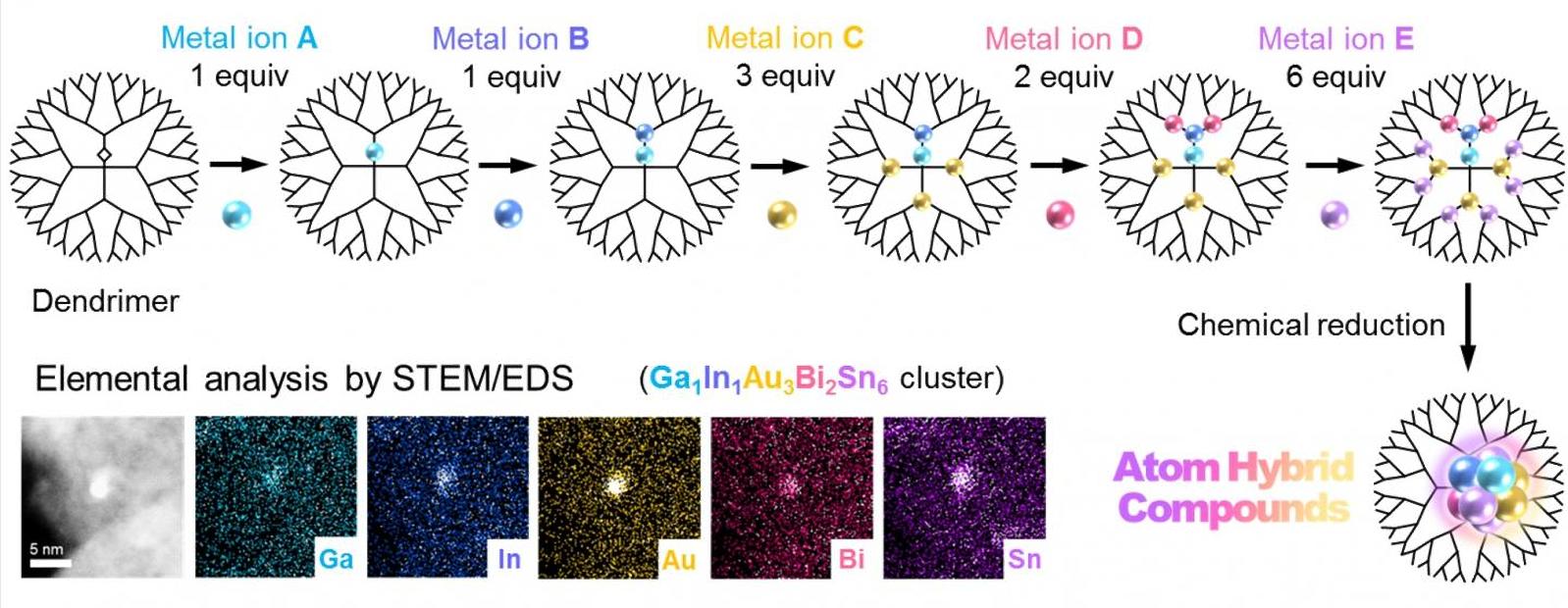
But up until now, no one had reported the multimetallic clusters blending with more than four metal elements due to the unfavorable separation of different metals. One thing that can be done to overcome this problem is the miniaturization of cluster sizes to one-nanometer scale, which then leads to the different metals to be blended in a small space. But there was no way to bring this idea to life.
A research team, including Takamasa Tsukamoto, Takane Imaoka, Kimihisa Yamamoto and colleagues, has developed an atom hybridization method, which has made the first-ever synthesis of multimetallic clusters possible, which consists of more than five metal elements with precise control of size and composition.
This method uses a dendrimer template that works as a tiny “scaffold” that allows the controlled accumulation of metal salts. After the precise uptake of all the various metals into the dendrimer, multimetallic clusters are achieved by chemical reduction. While a conventional method without the dendrimer leads to the enlargement of cluster sizes and separation of different metals stands in contrast.
The team was successful in demonstrating the formation of five-element clusters composed of gallium (Ga), indium (In), gold (Au), bismuth (Bi) and tin (Sn), as well as iron (Fe), palladium (Pd), rhodium (Rh), antimony (Sb) and copper (Cu), and also a six-element cluster consisting of Ga, In, Au, Bi, Sn and platinum (Pt). The research team also hinted at the possibility of making clusters composed of eight metals or more.